Alesis OSA1: Photonic Lipolysis to Target The Root Cause of Most OSA
First FDA-cleared, non-invasive device targeting the pathophysiologic mechanism of obstructive sleep apnea through localized adipose reduction for improved upper airway patency.
We manufacture the Alesis OSA1 in America for the sleep medicine community.
Schedule a Zoom MeetingAddressing the Anatomic Substrate of OSA
Recent MRI volumetric studies have established that tongue and parapharyngeal fat deposition represents a primary pathophysiologic mechanism in obstructive sleep apnea, independent of overall BMI.
- Wang et al., AJRCCM (2020): "Effect of Weight Loss on Upper Airway Anatomy and the Apnea-Hypopnea Index: The Importance of Tongue Fat" — MRI volumetric analysis demonstrates 32% weight loss produces 48% reduction in tongue fat (14,126mm³ to 7,337mm³) with corresponding 80% decrease in AHI (121 to 24.6).
- Parapharyngeal and retroglossal fat deposits shown to narrow pharyngeal airway diameter independent of BMI and total neck circumference.
- Bariatric surgery outcomes confirm AHI improvement occurs disproportionately early relative to total body weight reduction.
- Localized adipose reduction in upper airway structures represents mechanistic target independent of systemic weight loss.
- Clinical Trial NCT06949969: Randomized IRB study demonstrating 100% efficacy in submental fat reduction and 80% improvement rate in sleep apnea severity.
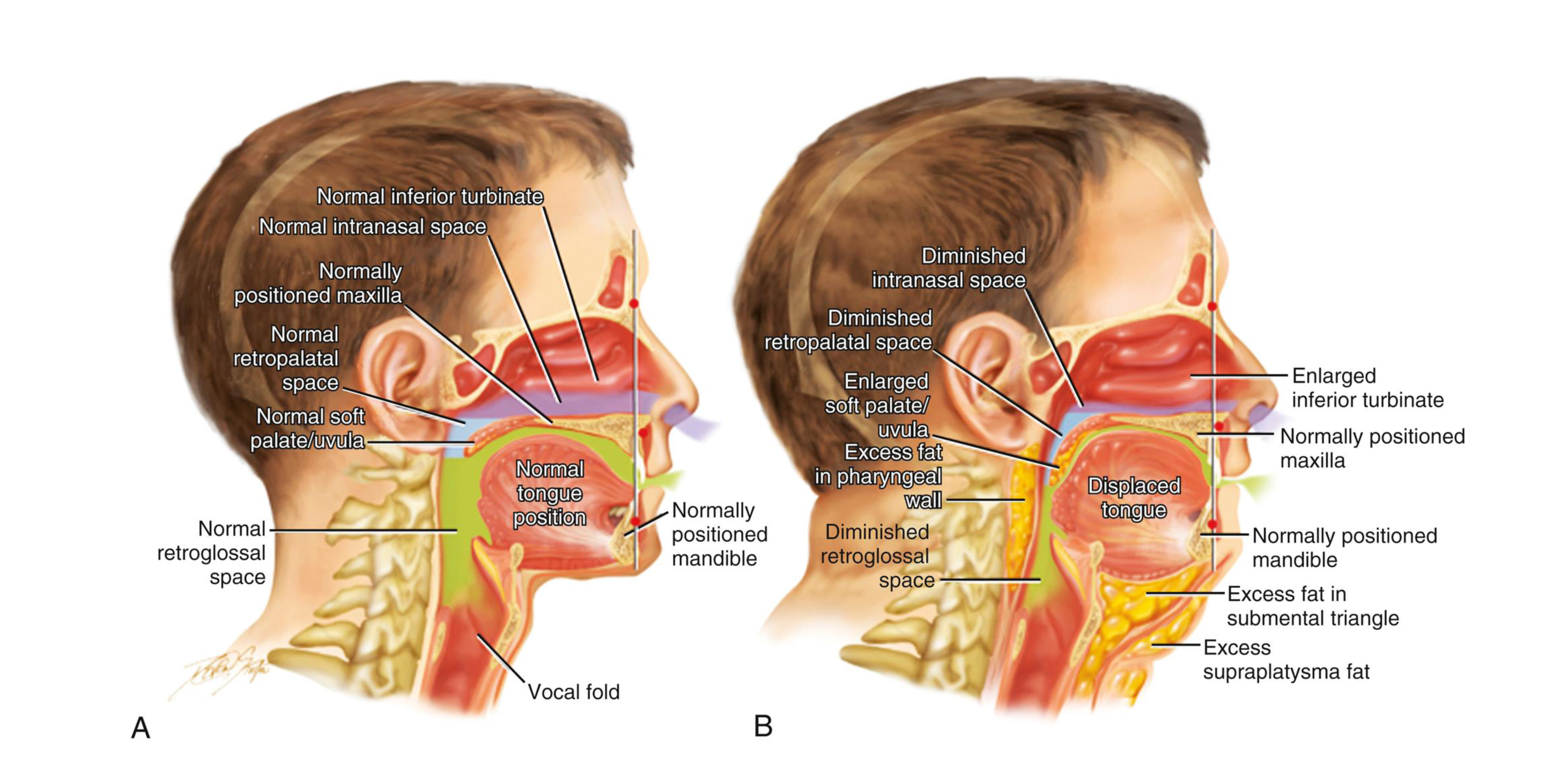
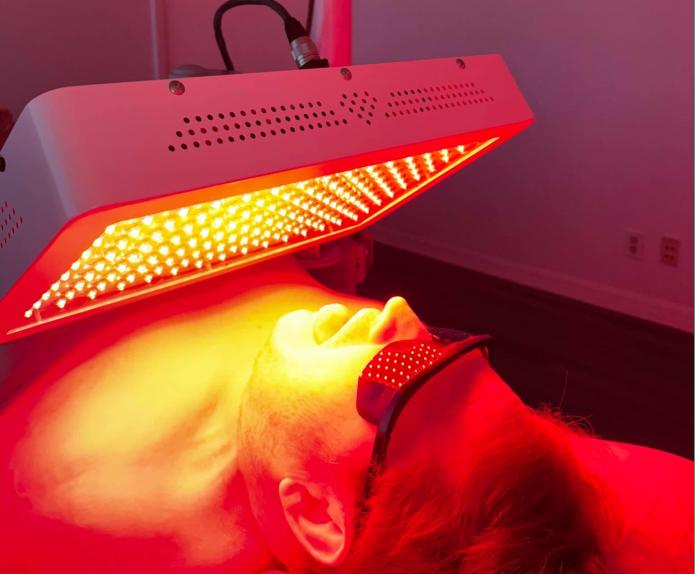
Mechanism of Action: Photonic Lipolysis
Patented high-powered non-laser photonic energy induces lipolysis through mitochondrial chromophore activation (cytochrome c oxidase), triggering cellular response mimicking famine state without thermal or destructive effects on adipocyte viability.
- Treatment Duration: 12 minutes per session, administered twice weekly for 3 weeks (total: 6 treatments)
- Anesthesia: None required; completely non-invasive outpatient procedure with no discomfort or downtime.
- Patient Selection: OSA diagnosis confirmed by polysomnography (AHI ≥5), overweight patients, CPAP-intolerant or seeking alternatives to PAP therapy.
- Selective Draping: Opaque draping available to protect facial areas where fat reduction is not desired (e.g., patients wanting airway improvement without facial volume loss).
- Contraindications: Pregnancy, active malignancy in treatment area, photosensitivity disorders, porphyria.
- Post-Treatment: Immediate return to all normal activities; zero recovery time or activity restrictions.
- Monitoring: Pre- and post-treatment polysomnography, neck circumference measurements, patient-reported outcomes.
OSA1 vs. Current Standard of Care
| Clinical Parameter | Alesis OSA1 | CPAP/BiPAP | Oral Appliance | UPPP Surgery |
|---|---|---|---|---|
| Addresses Root Cause | ✓ | ✗ | ✗ | Partial |
| Non-Invasive | ✓ | ✓ | ✓ | ✗ |
| No Nightly Compliance Required | ✓ | ✗ | ✗ | ✓ |
| Zero Recovery Time | ✓ | ✓ | ✓ | ✗ |
| Adverse Event Profile | None reported | Common (skin irritation, claustrophobia) |
Common (TMJ, dental) | Significant (pain, bleeding, infection) |
| Efficacy | Excellent | Excellent (if adherent) | Moderate | Variable |
| Patient Compliance | High | Poor (40%-50%discontinue) | Moderate | N/A |
Seamless Integration into Your Sleep Medicine Practice
Designed for efficient deployment within existing clinical workflows and comprehensive support infrastructure.
Request a consultation on the economics of the Alesis OSA1
Quality & Compliance
- K160880
- K150336
- K202361
Multiple 510(k) clearances establishing safety and efficacy across device iterations
- 11 Utility Patents
- 31 Countries Protected
- U.S. Patents: 9,044,595; 9,498,641; 9,808,314; 10,946,210; 63/074,790
Technology invented 2011 by Terry J. Ward, Sr., M.H.A.; OSA provisional patent granted February 2025
Made in USA
Manufactured by Photonica USA, LLC
Quality Assurance:
11 year safety record with zero adverse outcomes reported in clinical use
Partner with Alesis Medical
Join leading sleep medicine specialists offering advanced treatment options for OSA patients.
Direct physician inquiries welcomed: Contact Terry J. Ward, Sr., M.H.A., Managing Director & CEO